News for Industry
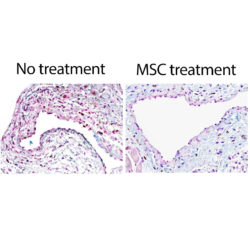
Mayo Clinic researchers are using stem cells from a patient’s FAT tissue to prevent inflammation and narrowing of vascular shunts used for hemodialysis. (Mayo Clinic News Network)
(Rochester, MN) Mayo Clinic researchers have addressed the narrowing of vascular shunts in patients with end-stage kidney disease by transplanting stem cells from the patient’s own fat tissue, which helped to prevent inflammation and narrowing of the vein.
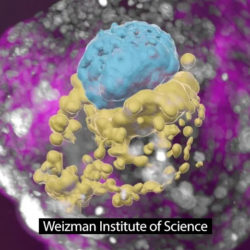
Scientists grew a whole model of a human embryo using stem cells, and without sperm or egg. (BBC.com)
(Rehovot, Israel) Researchers at the Weizmann Institute created an embryo model using only stem cells, no sperm or egg. Their Day 14 embryo looks textbook, even releasing pregnancy hormones which resulted in a positive pregnancy test in the lab.
The Florida Legislature approved a measure to improve access to stem cell therapies with strict requirements to ensure patient safety and ethical standards. (2025 Health News Florida)
(Tallahassee, FL) The Florida Legislature approved a measure to improve access to some stem cell therapies not yet by the FDA. Senate Bill 1768 authorizes stem cell treatments within scope of practice and with strict requirements to ensure patient safety and ethical standards.
Adipose tissue, otherwise known as fat, has the highest concentration of mesenchymal stem cells in the body. These stem cells produce and secrete extracellular vesicles. Collectively these are called the secretome. (sciencedirect.com)
(Palo Alto, CA) Stanford researchers have collaborated with scientists in Spain to program adipose (fat) stem cells to produce unique secretomes which focus on specific biological processes.

More funding needed for the fight: How stem cell transplant access can save young lives. (suidkaapforum.com)
(South Africa) South Africa’s public healthcare system is urging businesses, foundations, and individual donors to partner with their Department of Health to provide funding for stem cell research and therapy. They are encouraging this partnership to provide immediate financial support to “save lives now”. Response from the corporate sector has been positive but more is needed.

Florida Senator Jay Trumbull and Representative James Buchanan have filed similar bills to make stem cell therapy more available for patients in the US. (floridapolitics.com)
(Tallahassee, FL) New legislation could make stem cell therapies more accessible to patients in Florida and Senator Jay Trumbull and Representative James Buchanan have filed similar measures (SB 1768, HB 1617) to advance the cell therapy initiative.

Cartilage Regeneration Market Set to Reach $5.62 Billion by 2034, Driven by Advancements in Regenerative Medicine and Minimally Invasive Procedures (Research and Markets)
(Dublin, Ireland) The global cartilage regeneration market accounted for USD 1.16 billion in 2023 and is expected to reach USD 5.62 billion by 2034 with a CAGR of 15.43% during the forecast period 2024-2034.
UCLA scientists awarded $21.8 million in CIRM grants to advance novel stem-cell based therapies (UCLA Health)
Scientists at the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA have received $21.8 million in grants from the California Institute for Regenerative Medicine, the state’s stem cell agency, to develop and advance new stem cell-based treatments for neuropsychiatric disorders, a blood disorder and a neurodevelopmental condition.

Human endometrial stem cell-derived small extracellular vesicles enhance neurite outgrowth and peripheral nerve regeneration through activating the PI3K/AKT signaling pathway (Springer Nature)
Recently, various studies have demonstrated the therapeutic ability of exosomes, while the biological effect of human endometrial stem cell (hEnSC)-derived small EVs such as exosomes is still unclear…
